
In essence, just memorize the metalloids as a diagonal line on the periodic table and determine the other elements based on their orientation in relation to the metalloids. Those to the left side of this line of elements are metals and those to the right are nonmetals. If you look at the periodic table, these elements look to be in a diagonal orientation. Complete the definitions: Metalloid Malleable Luster Conductivity. They will use this to determine trends for metallic properties of the elements. Blue for metals, yellow for non-metals, and green for metalloids). Elements are further classified into metals, non-metals, and metalloids (semimetals). In other words, an element is the simplest form of matter. An element is a substance that cannot be broken down into any other substance. Insulators and Conductors 7.9K plays 4th - 5th 10 Qs. Introduction to Metals, Metalloids, and Nonmetals. Find other quizzes for Physics and more on Quizizz for free Skip to Content. The metalloids consist of boron, silicon, germanium, arsenic, antimony, tellurium, and polonium. There results will be color coded on a periodic table (e.g. Metals Nonmetals Metalloids quiz for 6th grade students. It can be used as an tour to the recurring table. They may have a metallic or dull appearance. These elements run diagonally across the Periodic Table. Boron, silicon, germanium, arsenic, antimony, and tellurium are the most commonly recognized metalloids. Hence, they are also known as semi-metals. One easy, general way to determine whether an element is a metal, metalloid, or nonmetal is to look at the periodic and to memorize the placement of the metalloids. This is adenine lab undertaking for one high schooling introductory chemistry laboratory. 18.3: Structure and General Properties of the Metalloids. Metalloids exhibit some properties of metals as well as non-metals. It arranges the 118 known elements into 7 rows ( known as periods ), 18 columns ( known as groups ) and several color. Metalloids are elements that are in between, and have properties that are in between those of metals and nonmetals. Nonmetals are on the upper right of the periodic table. Nonmetals on the other hand have a very high ionization energy and generally form anions (take on electrons). These are generally towards the left of the periodic table. Students will use the key to color the picture based on their answer. There are 10 questions for students to classify an element or identify location on the periodic table. 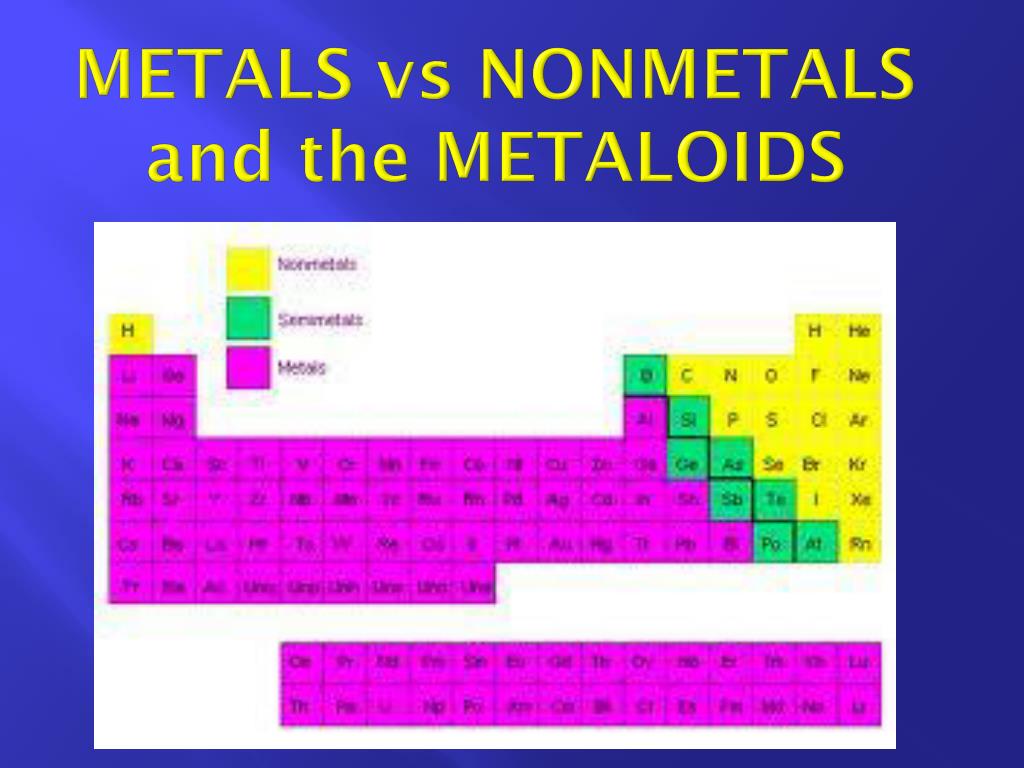
Metals have a low ionization energy, which allows them to be highly reactive and to form cations (lose electrons). Check for your students understanding of classifying metals, nonmetals, and metalloids with this science coloring page. You can tell whether most elements are metals, metalloids, or nonmetals based on their properties which you can determine through their placement on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
